
REE abundances and Eu isotope ratios in GSJ and NIST feldspar reference standards (JF-1, JF-2, SRM 70a, 70b and SRM 99a) using ICP-QMS and MC-ICP-MS
ABSTRACT
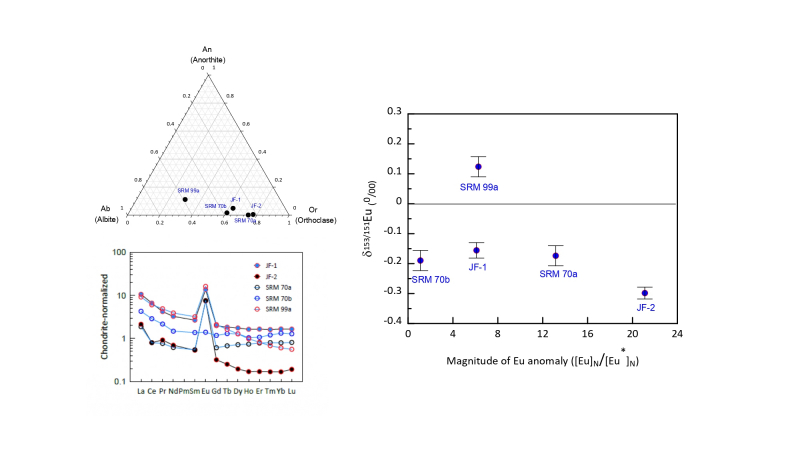
The rare earth element (REE) europium (Eu) exists in Eu2+ and Eu3+ states. The Eu2+ can be substituted for Ca2+ during plagioclase feldspar crystallization in reducing magmas to create an observable Eu anomaly in REE distribution patterns. Europium has two stable isotopes, 151Eu and 153Eu. Recent reports for Eu isotope ratios in igneous rocks indicate that Eu anomalies and isotope fractionation show good correlation, suggesting that Eu isotope fractionation appears due to feldspar crystallization during magma differentiation. Here, we report Eu isotope ratio and REE concentrations for five feldspar standard reference materials (SRMs) such as JF-1, JF-2, SRM 70a, SRM 70b, and SRM99a prepared by the Geological Survey of Japan (GSJ) and National Institute of Standards and Technology (NIST) of USA. This study is the first investigation of the Eu isotope variation in feldspars to test several hypotheses for the origin of Eu isotope fractionation in igneous rocks. The chemical compositions of four feldspar SRMs except SRM 99a indicate that they are close to the KAlSi3O8 end-member within the feldspar series. The SRM 99a meanwhile indicated oligoclase composition. Most feldspar SRMs showed chondrite-normalized patterns of light REE (LREE)-enrichment and heavy REE (HREE)-depletion or flat REE patterns with large positive Eu anomalies. This did not hold true for NIST SRM 70b, which contains small amounts of impurities. Results showed that potassium feldspar SRMs were enriched in the lighter Eu isotope (i.e., had negative of δ153Eu values), whereas the sodium feldspar SRM (NIST SRM 99a) were enriched in the heavier Eu isotope (i.e., had positive of δ153Eu values). Interpretation of Eu isotope ratios in igneous rocks should thus consider feldspar crystallization during magma differentiation.
KEYWORDS
Keywords: Eu isotope fractionation, Eu anomaly, feldspar SRMs, magma differentiation- Published : 2026
- Released on J-STAGE : -
- Received : 2025/05/21
- Accepted : 2026/04/03
- DOI : https://doi.org/10.2343/geochemj.GJ26007
- J-STAGE URL : https://www.jstage.jst.go.jp/article/geochemj/advpub/0/advpub_GJ26007/_article/-char/en
- J-Online ISSN: 1880-5973
- Print ISSN : 0016-7002
- ISSN-L : 0016-7002
All Issues
- Vol.60, 2026
- Vol.59, 2025
- Vol.58, 2024
- Vol.57, 2023
- Vol.56, 2022
- Vol.55, 2021
- Vol.54, 2020
- Vol.53, 2019
- Vol.52, 2018
- Vol.51, 2017
- Vol.50, 2016
- Vol.49, 2015
- Vol.48, 2014
- Vol.47, 2013
- Vol.46, 2012
- Vol.45, 2011
- Vol.44, 2010
- Vol.43, 2009
- Vol.42, 2008
- Vol.41, 2007
- Vol.40, 2006
- Vol.39, 2005
- Vol.38, 2004
- Vol.37, 2003
- Vol.36, 2002
- Vol.35, 2001
- Vol.34, 2000
- Vol.33, 1999
- Vol.32, 1998
- Vol.31, 1997
- Vol.30, 1996
- Vol.29, 1995
- Vol.28, 1994
- Vol.27, 1993
- Vol.26, 1992
- Vol.25, 1991
- Vol.24, 1990
- Vol.23, 1989
- Vol.22, 1988
- Vol.21, 1987
- Vol.20, 1986
- Vol.19, 1985-1986
- Vol.18, 1984
- Vol.17, 1983
- Vol.16, 1982
- Vol.15, 1981
- Vol.14, 1980
- Vol.13, 1979
- Vol.12, 1978
- Vol.11, 1977
- Vol.10, 1976
- Vol.9, 1975
- Vol.8, 1974
- Vol.7, 1973
- Vol.6, 1972-1973
- Vol.5, 1971
- Vol.4, 1970-1971
- Vol.3, 1969-1970
- Vol.2, 1968
- Vol.1, 1966-1967




